- Enfermedades del Corazón en Adultos
- Enfermedad de la válvula aórtica
- Enfermedad de las Arterias Coronarias
- Enfermedades de la Válvula Mitral
- Enfermedad de la Válvula Tricúspide
- Trastornos del Ritmo Cardíaco
- Aneurisma de la Aorta Torácica
- Disección Aórtica
- El Dispositivo de Asistencia Ventricular Izquierdo (DAVI)
- Fibrilación Auricular (FA)
- OMEC VA
- La insuficiencia cardíaca
- Trasplante de corazón en adultos
Diseases of the arteries, valves, and aorta, as well as cardiac rhythm disturbances - Pediátrica y Cardiopatías Congénitas
- Comunicación interauricular
- Comunicación interventricular
- Tetralogía de Fallot
- Defecto del canal auriculoventricular
- Transposición de las grandes arterias
- Coartación de la aorta
- Tronco arterial común
- Defectos de ventrículo único
- Persistencia del conducto arterioso
- Trasplante de corazón en pacientes pediátricos
- Trasplante de pulmón en pacientes pediátricos
Heart abnormalities that are present at birth in children, as well as in adults - Pulmón, esófago, y otras enfermedades del pechoDiseases of the lung, esophagus, and chest wall
- ProceduresCommon surgical procedures of the heart, lungs, and esophagus
- Pre- y Post- Cuidado Operativo
- Antes de la cirugía de corazón
- El día de la cirugía de corazón
- Después de la cirugía de corazón
- Antes de la cirugía de cáncer de pulmón
- El día de la cirugía de cáncer de pulmón
- Luego de la cirugía para el cáncer de pulmón
- Antes de la cirugía para cardiopatías congénitas
- El día de la cirugía para cardiopatías congénitas
- Después de la cirugía para cardiopatías congénitas
How to prepare for and recover from your surgery
For patients with early-staged lung cancer, surgery to remove the tumor and lymph nodes in the area has been a long-accepted strategy for cure. If you or a family member has been recently diagnosed with lung cancer that was caught early, it is understandable that you might be eager to get it removed, without any delays related to biopsies or genetic evaluations.
For patients with more advanced disease, such as those individuals with larger tumors or with lymph node involvement, treatment usually requires a combination of therapies—and while surgery might be part of the treatment, it might be accompanied by medications to help treat the disease and reduce the risk of recurrence. In these situations, likewise, it’s normal to be eager to get treatment started, rather than wait for results of biopsies and molecular markers.
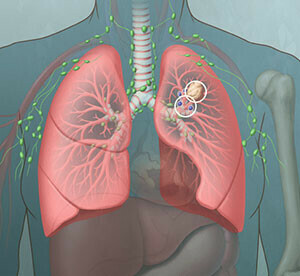
Why, then, is it so important to perform tumor marker testing before we embark upon a treatment strategy?
Well, it’s important to first understand the relationship between these tumor markers and the way that a tumor grows. Specific abnormalities in a tumor’s cells, related to its DNA and the proteins that the cell makes, contribute to the tumor cells growing faster than normal, forming a mass, and eventually help those tumor cells to spread and invade other sites in the body.
In the past, the main medications that we had for lung cancer were chemotherapy agents, and, even in order to pick the most effective chemotherapy drug for a given patient, it’s best to first know what the tumor looks like under a microscope. This emphasizes the importance of a biopsy before starting treatment.
And now, in recent years, we’ve seen lots of new drugs as well as new situations in which we can use them. Many of these newer medications that are available for lung cancer, such as immunotherapy or targeted therapies, are able to stop tumors from growing by addressing the specific abnormalities in the tumor’s DNA or the proteins it makes (protein expression) to kill the cancer cells while causing minimal harm to the normal, healthy cells of the body.
For example, we know that immunotherapy may be more effective in patients with certain proteins expressed from their tumor cells. Immune-checkpoint inhibitors are a type of immunotherapy drugs that tend to have the best results in patients whose tumors have high expression of a protein called PD-L1. Moreover, we now have recent research that shows us that immunotherapy can be helpful before surgery for some patients, and after surgery for others.
Similarly, targeted therapies tend to work best on patients who have tumors with genetic mutations specifically addressed by that drug. EGFR is a common mutation in lung cancer, and we now have drugs called EGFR tyrosine kinase inhibitors that work quite well for patients with that particular genetic mutation. Likewise, we have drugs that are targeted for patients with abnormalities in the ALK gene. And in recent years, we’ve gained important knowledge telling us that we can improve cancer outcomes by giving these types of targeted therapies after surgery, even for patients who have achieved a complete surgical resection.
It’s estimated that 1 in 3 patients may have an abnormality in their cells’ DNA or proteins that could be directly treated with the drugs that we have today—and it’s very likely that even more abnormalities will be able to be treated with a tailored medication in the future!
But what if the tumor can just be removed?
Historically, we used to think that tailored medications such as targeted and immunotherapy were only relevant for patients with advanced or recurrent disease. However, we have had a number of recently completed and ongoing trials that are showing benefit from these novel drugs in the preoperative and postoperative setting.
Thus, it’s important for us to know details about an individual’s molecular markers for several reasons:
- It may tell us if you’d benefit from receiving an individualized drug before surgery.
- It may give us information about the need for receiving a drug after surgery to reduce risk of recurrence.
- Even if no medication is needed at this time, it might give us some clues about how to best treat the tumor if it were to come back in the future.
And what if you have already had a complete resection—is there still a need for molecular profiling?
Absolutely! Having a successful operation is a terrific milestone, and it is a just cause for celebration for patients and their families. After getting through a procedure and its associated potential challenges, discomfort, and disruption to daily life, being cancer-free is a wonderful reason to rejoice. The risk of cancer recurring will never be zero, but we can often work hard to prevent it from coming back by treating with appropriate medications when indicated. In addition, we strongly advocate for obtaining routine screening scans, so that if something unexpected is present, we can find it early, and we can use previous molecular markers to promptly target any recurrent disease.
We haven’t yet developed drugs for all of the molecular abnormalities that we have identified in lung cancer, and we certainly have not yet found all of the possible genetic abnormalities that can drive lung cancer. But our knowledge is rapidly growing, and one thing is for sure: detailed knowledge about an individual’s tumor cells is the most powerful weapon against their cancer.


